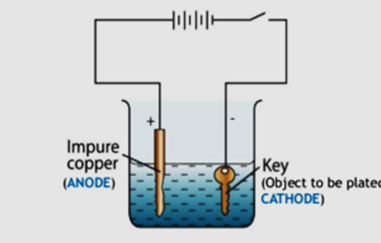
With the help of a labelled diagram, describe briefly how an iron key can be electroplated with copper.
We need two electrodes made from different conducting materials (here copper and iron as anode and cathode respectively), an electrolyte, and an electricity supply. The iron key will be taken as the cathode (negative electrode ) and the copper to be electroplated on iron key to be made the anode (positive electrode). When we open the electricity, then copper ions are deposited on the negative electrode and in this way copper is electroplated on the iron key.
46