50 cal of heat should be supplied to take a system from the state A to the state B through the path ACB as shown in the figure. Find the quantity of heat to be supplied to take it from A to B via ADB.
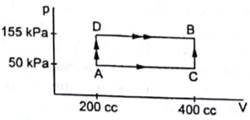
From graph we can write
VA =VD = 200cc=200×10-6m3
VB=VC = 400cc= 400×10-6m3
PB=PD=155kPa= 155×103Pa
PA=PC=50kPa=50×103Pa
Given
Heat absorbed in process ABC ΔQ1=50cal= 50×4.2 J=210J
Let heat absorbed into the system during process ADC =ΔQ2
We know that work done by the gas is given as
ΔW=PΔV
Work done in path ACB WACB =ΔW1= WAC+WBC
ΔW1=PA(VC-VA) + 0 (WBC=0 because VB=VC)
=50×103×(400-200)×10-6
=10J
Work done in path ADB WADB=ΔW2=WAD + WDB
ΔW2 = PB (VD-VB) +0 (∵ WAD=0 because VA=VD)
=155×103×(400-200)×10-6
=31 J
Now initial point A and final point C is the same for both the processes is the same. So, change in internal energy will be the same for both the process, as internal energy is a state function independent of the path taken.
Therefore,
ΔU1=ΔU2=ΔU ……(i)
From first law of thermodynamics, we know that,
ΔQ=ΔU+ΔW
Where ΔQ=heat supplied to the system
ΔU=change in internal energy
ΔW=work done by the system
Using first law of thermodynamics for process ABC
ΔQ1=ΔU1+ΔW1
ΔU1=ΔQ1-ΔW1=210-10=200J
Using first law of thermodynamics for process adc
ΔQ2=ΔU2+ΔW2
=ΔU1+ΔW2 (from (i))
=200+31=231J.
∴ heat supplied to the system during process ADC=231J.