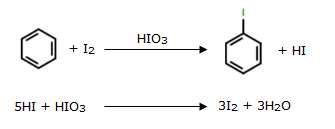
Aryl chlorides and bromides can be easily prepared by electrophilic substitution of arenes with chlorine and bromine respectively in the presence of Lewis acid catalysts. But why does preparation of aryl iodides requires presence of an oxidising agent?
Iodination of arenes is reversible in nature due to formation of HI. To move the reaction forward, an oxidizing agent like HNO3 or HIO4 oxidises HI, thus stabilizing the product.
1