The molar heat capacity for the process shown in the figure is
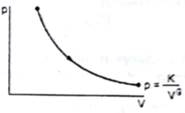
Given : ![]() which gives us
which gives us
pVg = K, where p = pressure, V = volume, and g and K are constants. This represents an adiabatic process.
In an adiabatic process, Q(heat exchanged) = 0.
Now we know that Q = nCdT,
where n = number of moles, C = specific molar heat capacity and dT = rise in temperature.
Since Q = 0, we get C = 0.
Hence, the correct option is (d).
1