An ideal gas (CP/CV = γ) is taken through a process in which the pressure and the volume vary as p = αVb. Find the value of b for which the specific heat capacity in the process is zero.
Given:
p = aVb
p = pressure, V = volume, a and b are constants.
Formula used:
We know, Q = U + ഽpdV from first law of thermodynamics,
where Q = change in heat, U = change in internal energy and ഽpdV = W = total work done, p = pressure, V = volume.
Since Q = nCdT, and U = nCvdT, we get
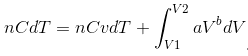 … (ii), n = no. of moles, C = specific
… (ii), n = no. of moles, C = specific
heat capacity, and Cv = specific heat capacity at constant volume,
dT = change in temperature,
Since specific heat capacity is 0(given),
![]() …(iii)
…(iii)
(after integration of 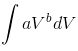 from V1 to V2)
from V1 to V2)
Now, from equation of state, PV = nRT,
Where
P = pressure,
V = volume,
n = number of moles,
R = universal gas constant,
T = temperature.
Substituting p = aVb from (i):
aVb+1 = nRT
=> ![]() … (iv)
… (iv)
Substituting (iv) in (iii),
![]()
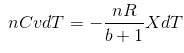 (Since (T2 - T1) = dT)
(Since (T2 - T1) = dT)
=> 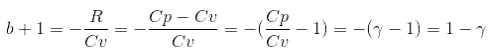
=> b = -