What is meant by refining of a metal ? Name the most widely used method for the refining of impure metals obtained by various reduction processes. Describe this method with the help of a labelled diagram by taking the example of any metal.
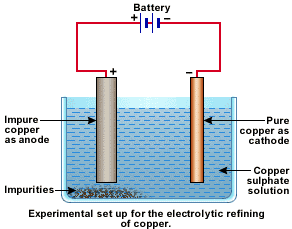
A small electric current is passed through the cell. Atoms from the anode enter the electrolyte. The copper from the anode gets converted into copper sulphide. An equal number of copper atoms from the solution get deposited on the cathode. This is to keep the concentration of the solution constant. Impurities from the anode block either remain in solution or collect below the anode, as they are unable to displace copper from the sulphate Solution. The insoluble impurities remain in the electrolyte and are called anode mud.
Copper sulphate solution contains ions of Cu++ and SO4--. The following reactions take place at the anode and cathode when an electric current is passed.
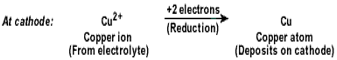

Pure copper is scraped or removed from the cathode. Anode becomes thinner as the electrolysis process proceeds. Some important metals like gold and silver are present in the anode mud. These can be recovered separately.