Write a balanced chemical equation for process of photosynthesis.
Photosynthesis reaction: CO2 + H2O![]() C6H12O6 + O2 + H2O
C6H12O6 + O2 + H2O
Balanced equation: 6CO2 + 12H2O![]() C6H12O6 + 6O2 + 6H2O
C6H12O6 + 6O2 + 6H2O
Step 1: Write the unbalanced equation
CO2 + H2O![]() C6H12O6 + O2 + H2O
C6H12O6 + O2 + H2O
Step 2: Compare the number of atoms of reactants with the number of atoms of products.
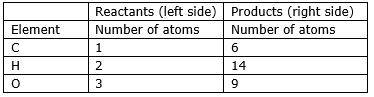
Step 3: Now, first we consider the element having unequal no. of atoms on both sides. Thus, first let us consider carbon atom. If we multiply 6 in the reactant (in CO2), we will get the equal number of atoms as in product (in C6H12O6)
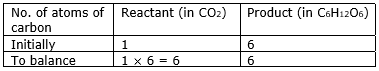
Step 4: Write the resulting equation:
6CO2 + H2O![]() C6H12O6 + O2 + H2O
C6H12O6 + O2 + H2O
Step 5: Now check whether the equation is balanced or not by comparing the atoms
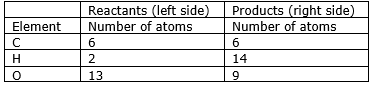
We find that the equation is not balanced yet. As the number of
Hydrogen and oxygen atoms are unequal on the two sides.
first balance the hydrogen atom.
Step 6: If we multiply 12 in the reactant (in H2O) and 6 in the product (in H2O), we will get the equal number of hydrogen atoms both sides.

Step 7: Write the resulting equation:
6CO2 + 12H2O![]() C6H12O6 + O2 + 6H2O
C6H12O6 + O2 + 6H2O
Step 8: Now check whether the equation is balanced or not by comparing the atoms.
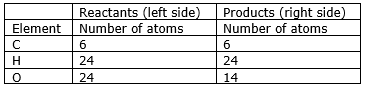
We find that the equation is not balanced yet. As the number of oxygen atoms are unequal on the two sides.
Step 9: Now, we consider oxygen atoms. If we multiply 6 in the product (in O2), we will get the equal number of atoms as in reactants (in 6CO2 and 12H2O)

Step 10: Write the resulting equation:
6CO2 + 12H2O![]() C6H12O6 + 6O2 + 6H2O
C6H12O6 + 6O2 + 6H2O
We find that the equation is balanced now.
Step 11: Write down the final balanced equation:
6CO2 + 12H2O![]() C6H12O6 + 6O2 + 6H2O
C6H12O6 + 6O2 + 6H2O