Write the molecular formula of i. ethane and ii. Ethanoic acid.
(i) The prefix “eth-” corresponds to 2 carbon atoms, and the suffix “-ane” tells us that the compound is an alkane.
Ethane is a saturated hydrocarbon (alkane) containing 2 carbon atoms. The general molecular formula of alkanes is CnH2n+2, where n is the number of carbon atoms. For n=2, we get the molecular formula of ethane which is C2H2x2+2 or C2H6.
(ii) The prefix “eth-” corresponds to 2 carbon atoms, and the suffix “-oic acid” tells us that the compound is a carboxylic acid.
Ethanoic acid is a carboxylic acid containing 2 carbon atoms. One of the carbon atoms is included in the carboxylic acid functional group (-COOH or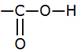 ) and is attached to the carbon atom. The remaining valency is satisfied by hydrogen atoms.
) and is attached to the carbon atom. The remaining valency is satisfied by hydrogen atoms.
Therefore, the molecular formula is C2H4O2 (CH3COOH).